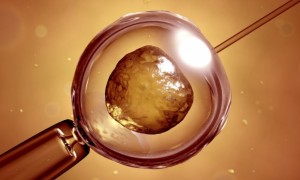
Vanna Belton from Baltimore has been blind for more than five years, but after undergoing surgery where stem cells extracted from her bone marrow were injected into her right eye’s retina and left eye’s optic nerve, she has regained some of her sight.
“When I realised I could see the license plates, we started walking around the neighbourhood reading them,” she told the Baltimore Sun, adding that for the first time since 2009, she’s been able to navigate her way around without a cane.
While Belton’s recovery is certainly remarkable, it’s also incredibly perplexing. The doctor who performed the stem cell treatment, ophthalmologist Jeffrey N. Weiss, cut so many corners to get her and 277 other patients into surgery, even he can’t explain why it works.
“Weiss is not following the usual steps of clinical studies,” Meredith Cohn reports for the Baltimore Sun. “Among other things, he didn’t test his treatment theories first on lab animals or using computer models, or randomise his trials by using either stem cells or placebos in study participants. He didn’t test the procedure for safety on a small group before moving to a larger trial.”
Essentially, the ‘traditional route’ of lab-to-animals-to-clinical-trials takes too long, Weiss says, so he figured out how to game the system and start testing his new treatment on his own terms.
All he had to do was register his human trials with the US National Institutes of Health (NIH), which usually requires approval from the Food and Drug Administration (FDA) before any new or established drugs can be tested on humans.
The catch is that stem cells aren’t classified as a drug – they’re extracted from a patient’s body, go through very little processing, and are used on that same patient, so Weiss was able to bypass FDA approval to register his NIH trials. He did need approval and oversight from an ethics review panel, and got his from the International Cellular Medicine Society, an independent, pro-stem cell therapy group based in Nevada.
So Weiss, who is practicing independently of any university or research institution, is now free to charge around US$20,000 per patient to perform his stem cell treatment, and offers no promises of a cure to his patients.
But according to Cohn, Weiss reports that 60 percent of his 278 patients – who have gone blind because of diseases such as macular degeneration and glaucoma – have regained some sight following the procedure. He’s published a case study on Belton in the journal Neural Regeneration Research.
Belton lost her sight in a matter of weeks in 2009 due to a severe case of optic neuritis – inflammation of an optic nerve that causes progressively blurred vision because the nerve could no longer communicate with the brain. Until recently, dark shadows were the only thing should could make out, and she used a cane and various technological devices to help her function.
After undergoing Weiss’s 4.5-hour surgical procedure, in which stem cells extracted from bone marrow in her hip were injected into a different area in each eye, she gradually regained some of her sight. She can now read menus and street signs, get around without her cane, and has seen her wife’s face for the first time in five years.
We should point out that the procedure is by no means a cure – Belton is still considered to be legally blind, and rather than seeing everything now, she can only make out ‘islands’ of sight in a sea of nothing, Cohn reports. But you’d be hard-pressed to find someone who wouldn’t pay $20K to get at least some of their sight back.
The problem – likely confounded by the fact that Weiss cut so many corners – is that no one can explain how the treatment actually works.
For example, how does he explain the fact that Belton has regained sight in both her eyes, despite the fact that the stem cells were injected into her right eye’s retina and left eye’s optic nerve? Does that mean it doesn’t matter where the stem cells are injected into the eye, and if that’s the case, how does that even work?
Weiss says he or someone else will figure that out eventually, but previous research points to two possibilities – either the injected stem cells revive malfunctioning stem cells in the eye, or they replace them.
“We didn’t know how penicillin worked for many years, but it saved many lives in the meantime,” Weiss told the Baltimore Sun. “It is hubris to think that something can’t work until you understand how it does. … It is more important what the patient sees, not what I see.”
Right now, there are a number of clinical studies in the US that are also investigating the potential of stem cell treatments, but the earliest results of those are not expected until later this year. And late last year, we reported onthe first person in the UK to receive stem cell treatment for their blindness (the results are still yet to be announced), so we are definitely going to hear much more about this in the near future.
What other researchers in the field are really hoping for is a proper report on Weiss’s 278 patients, so it can be peer-reviewed and scoured for clues that would explain the mechanism that’s restoring sight to some, but not others.
Another drawback of pursuing results before fully understanding the science is that it’s not clear if further treatment will hurt or hinder Belton and Weiss’s other patients’ progress. Belton’s scheduled to receive more stem cell injections, but has been warned that while it could give her more sight, it could also take away the sight that’s been restored.
“I’m happy to be a guinea pig,” she told Cohn.
Source: Sciencealert








































































































































































































































![[Video] Chicago Police Officers Caught On Video Telling Two Black Men "We Kill Mother F**kers"](https://earhustle411.com/wp-content/uploads/2018/07/evil-cop-3-300x180.jpg)
![[Video] Chicago Police Officers Caught On Video Telling Two Black Men "We Kill Mother F**kers"](https://earhustle411.com/wp-content/uploads/2018/07/evil-cop-3-80x80.jpg)












![[Video] White Woman Calls The Cops On Black Real Estate Investor, Cops Threaten To Arrest Her For Harassing Him](https://earhustle411.com/wp-content/uploads/2018/05/nosy-neighbor-300x180.png)
![[Video] White Woman Calls The Cops On Black Real Estate Investor, Cops Threaten To Arrest Her For Harassing Him](https://earhustle411.com/wp-content/uploads/2018/05/nosy-neighbor-80x80.png)


![White Scientist Says The Black Community Is Being Targeted By The Medical System, They Are Deliberatly Being Poisoned [Video]](https://earhustle411.com/wp-content/uploads/2016/05/mike-adams-300x180.jpg)
![White Scientist Says The Black Community Is Being Targeted By The Medical System, They Are Deliberatly Being Poisoned [Video]](https://earhustle411.com/wp-content/uploads/2016/05/mike-adams-80x80.jpg)







![Teenage Girl Shot In Her Stomach Three Times But Took Time To Post To Facebook [ Video]](https://earhustle411.com/wp-content/uploads/2016/02/Gangster-chick-300x180.jpg)
![Teenage Girl Shot In Her Stomach Three Times But Took Time To Post To Facebook [ Video]](https://earhustle411.com/wp-content/uploads/2016/02/Gangster-chick-80x80.jpg)